
Sodium as the positive spectator ion and NH2 minus as the negative and reactive Anion. You will also see something like Na NH2 which ones again has Sodium as our positive spectator ion and NH2 minus as our negative Ion. Common examples include something like NaOCH3 which is a positive Sodium spectator and Methoxide is your anion. You’ll typically find the group 1 elements at the start of a larger molecule written out as a neutral compound but you should recognize them as a cation and therefore whatever follows is likely an anion.įor example, anytime you see Lithium, Sodium or Potassium followed by something that has an Oxygen or Nitrogen we’re likely looking at an ionic compound. You will find group 1 elements often forming a cation or positive ion. Since the quickest way to reach a full octet is to give away that valence electron. Looking at the table, in group 1 all the elements have one valence electron. I go through the idea of Periodic Table and Trends In another video which you can find on my website at. If you look at the periodic table, you should be able to recognize the atoms that are likely to form a covalent bond based on the number of valence electrons and where they’re located. In Organic Chemistry it’s more a concept of recognizing when these bonds take place. In General Chemistry you learn how to calculate the difference in electronegativity and to recognize when the difference is greater than two, you’ll have an ionic bond or have one atom steal from another. This is easy to see in atoms like Sodium which has one valence electrons and when lost gets a net charge of plus 1 or Chlorine which has 7 valence electrons and when it gains one more gets a net negative charge. 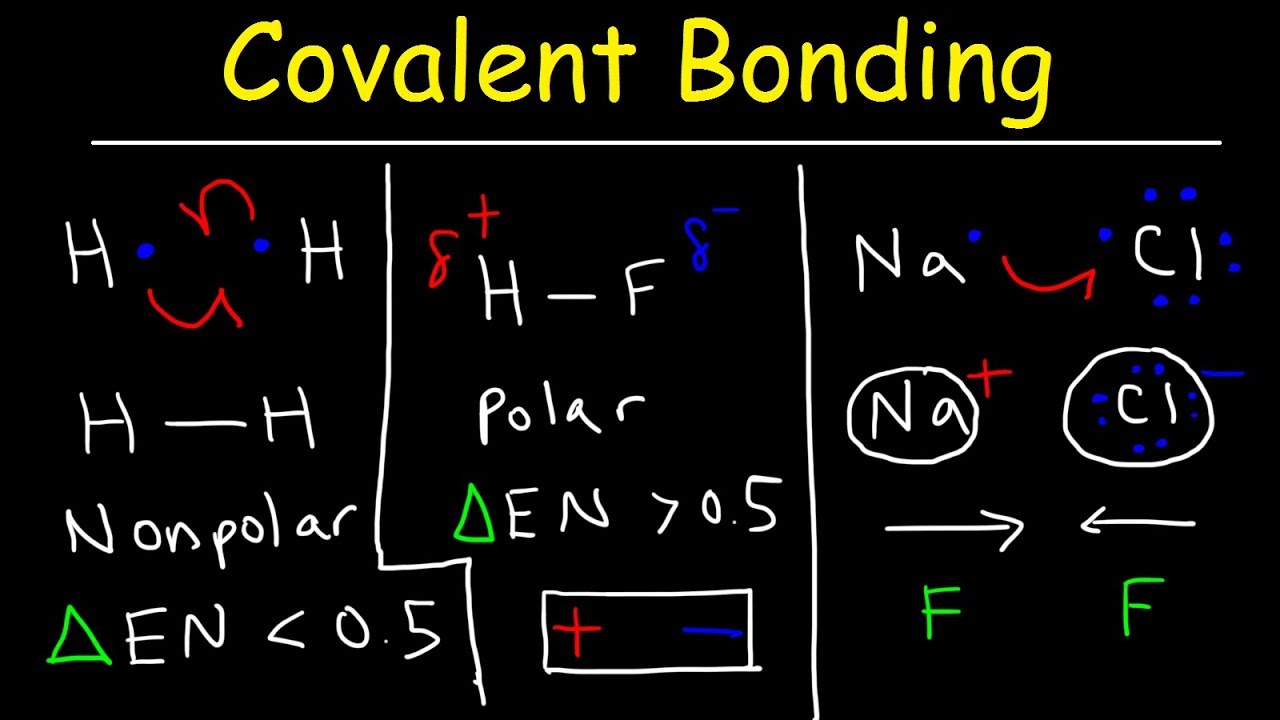
An Ion is an atom or molecule that gained or lost an electron and since the number of protons and electrons are not equal to each other we’ll have a net negative or net positive charge. When you cover this in General Chemistry you look at the idea of the octet rule and how atoms that do not have a complete octet will interact with other atoms to help each other reach that full valence shell of electrons. Leah here from and in this Orgo basics video we’ll talk about bonding as it’ll show up in your Organic Chemistry course including Ionic Bonding, Polar covalent and non polar covalent. Note that these transactions can normally only take place simultaneously: in order for a sodium atom to lose an electron, it must be in the presence of a suitable recipient like a chlorine atom.Below is the transcript of my tutorial video in Ionic, Polar Covalent and Non-Polar Covalent Bonding. Because the number of electrons is no longer equal to the number of protons, each is now an ion and has a +1 (sodium cation) or –1 (chloride anion) charge. Both ions now satisfy the octet rule and have complete outermost shells. 
In this example, sodium will donate its one electron to empty its shell, and chlorine will accept that electron to fill its shell. Therefore, it tends to gain an electron to create an ion with 17 protons, 17 neutrons, and 18 electrons, giving it a net negative (–1) charge. Again, it is more energy-efficient for chlorine to gain one electron than to lose seven. Chlorine (Cl) in its lowest energy state (called the ground state) has seven electrons in its outer shell. If sodium loses an electron, it now has 11 protons, 11 neutrons, and only 10 electrons, leaving it with an overall charge of +1. It takes less energy for sodium to donate that one electron than it does to accept seven more electrons to fill the outer shell. As Figure 1 illustrates, sodium (Na) only has one electron in its outer electron shell. This movement of electrons from one element to another is referred to as electron transfer. Anions are designated by their elemental name being altered to end in “-ide”: the anion of chlorine is called chloride, and the anion of sulfur is called sulfide, for example. Negative ions are formed by gaining electrons and are called anions. Cations are positive ions that are formed by losing electrons. Because the number of electrons does not equal the number of protons, each ion has a net charge. This fills their outermost electron shell and makes them energetically more stable. Some atoms are more stable when they gain or lose an electron (or possibly two) and form ions. Describe the characteristics of ionic bonds and identify common ions.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
